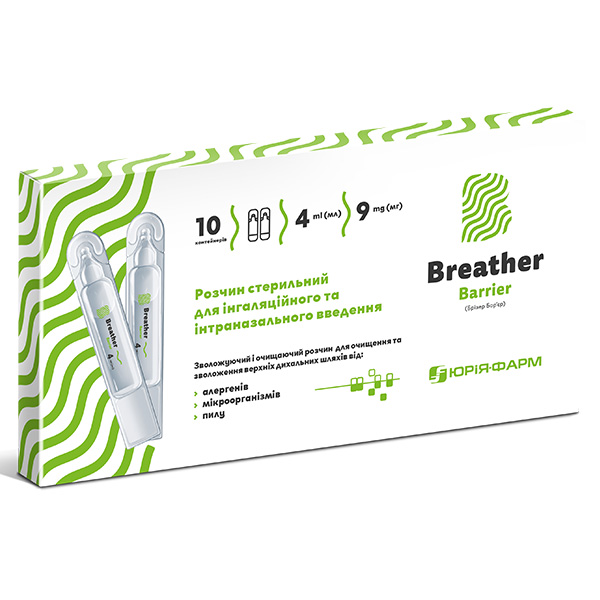
Breather Barrier
Breather Barrier is a special solution for “breathering” procedure used to moisturize and clean the mucous membrane of the upper airways from allergens, dust and microorganisms (as well as for use in the Breather Airox inhaler system).
Indications:
- Hygiene care, moistening and cleaning the mucous membranes of upper airways in the rooms with dry air caused by conditioners (including automobile conditioners), heaters and radiators.
- Cleaning the mucous membrane of upper airways from allergens during flowering period and dust containing in the polluted air.
- Patients with diseases of nasal mucosa and paranasal sinuses accompanied by dryness of mucous membrane and mucus formation (atrophic, allergic, drug-induced rhinitis).
- After surgical interventions in nasal cavity and nasal sinuses.
For breathering with Breezer Barrier solution, the Breather AirOx Mesh VP-M3 nebulizer is used.
How supplied
Sales markets
Ukraine
Instruction
INSTRUCTIONS FOR USE
of medical device
Sterile solution for inhalation and intranasal administration
BREATHER BARRIER
Composition
4 ml of solution contains: sodium chloride – 9 mg/ml, water for injection.
Contents of packaging
4 ml in polymer container, 10 containers in a cardboard package.
Description
BREATHER BARRIER is a sterile solution for inhalation and intranasal administration. It is a natural isotonic saline solution, concentration of which is adapted to be compatible with human body liquids. Solution is used for moisturising and cleaning the upper respiratory tract from allergens, microorganisms and dust, as well as using an inhalation system.
Intended purpose
BREATHER BARRIER is intended to moisturize and clean the upper respiratory tract.
Indications
Hygiene care, moisturizing and cleaning the mucous membrane of the upper respiratory tract in premises with dry air caused by air conditioners (including car air conditioners), heaters, central heating radiators, during air flights, from allergens in the period of blossom and dust in polluted air. To be used in patients with diseases of nasal mucosa and paranasal sinuses, accompanied by dryness of the mucous membrane or by mucus formation (atrophic, allergic, infectious, drug-induced rhinitis).
As an adjunctive agent while usage of local vasoconstrictors.
After nasal cavity and maxillary sinuses surgeries.
Furthermore, it can be used for dilution of inhalation medicinal products according to the instruction of the manufacturer.
Contraindications
Individual hypersensitivity to the components of the device is available.
Target group (population)
BREATHER BARRIER is intended for use in infants, children and adults with the need to moisturize and clean the upper respiratory tract.
Method of use
Administration through the inhalation system
Inhalation of solution BREATHER BARRIER is to be carried out through the inhalation system applying a special face mask, mouthpiece, or nasal cannula.
Familiarize yourself with the instructions for use of the inhalation system before the procedure. Follow the operating instructions for the inhalation system model you are using.
- Prepare the inhalation system for use.
- Open the foil bag and separate the single-dose container. Make sure the container is not damaged.
- Shake the separated container. Leave the other containers in the foil bag and put them into the cardboard box. Do not remove the container from a bag unless necessary.
- Holding the container by its upper edge, rotate the other edge to open the container.
- Insert the container into the inhalation system with the open edge down and give a slight press. Make sure that all the solution flew into a tank of device.
- Close the tank of inhalation system and use it as intended.
- Wash the inhalation system after usage, dispose the solution residues.
Intranasal administration
Adults (over 18 years old) – 2 drops; children above 1 year of age – 1‒2 drops; infants – 1 drop into each nasal passage 3‒4 times a day for therapeutic purposes, for hygiene – 1‒4 times a day.
Apply up to 4 weeks.
Method of administration for adults: prior to procedure it is required to wash the hands with soap and release the nasal passages carefully from secretion with a rapid forceful exhalation through the nose. To prevent the solution outflow, it is required to lie down or sit down, throw back the head and then drip the solution. While drops instillation into the right nasal passage, the head should be thrown back slightly and inclined leftward and vice versa, the head should be inclined rightward while drops instillation into the left nasal passage. After drops instillation, it is advisable to stay in a supine position with a head thrown back for 2 minutes, then release the nasal passages from the liquefied secretion. Wipe the solution residues on the face with a napkin.
Method of administration for children: prior to procedure it is required to wash the hands with soap and release the nasal passages of child from secretion carefully. After cleaning the nose from secretion, put the drops into each nasal passage. While drops instillation into the right nasal passage, the child’s head should be thrown back slightly and inclined leftward and vice versa, the child’s head should be inclined rightward while drops instillation into the left nasal passage. After drops instillation in children, it is advisable to stay in a supine position with a head thrown back for 2 minutes, then help the child to sit down and release the nasal passages from the liquefied secretion. Wipe the solution residues on the face with a napkin.
For dilution of inhalation medicinal products use 2‒4 ml of solution. Use for dilution only those medicinal products the instruction of which indicates the possibility of dilution with 0.9% saline sodium chloride.
Adverse reactions
In persons with individual intolerance of the components of medical device hypersensitivity reactions may occur.
In the event of any adverse reactions, the use of the medical device should be discontinued immediately and the physician and the manufacturer should be informed.
Limitations, precautions and warnings
- Solution is intended to be used only via inhalation and intranasal administration. Apply the device as per the instruction for medical use. Consult your physician before using if you have questions.
- Use in children under the supervision of adults.
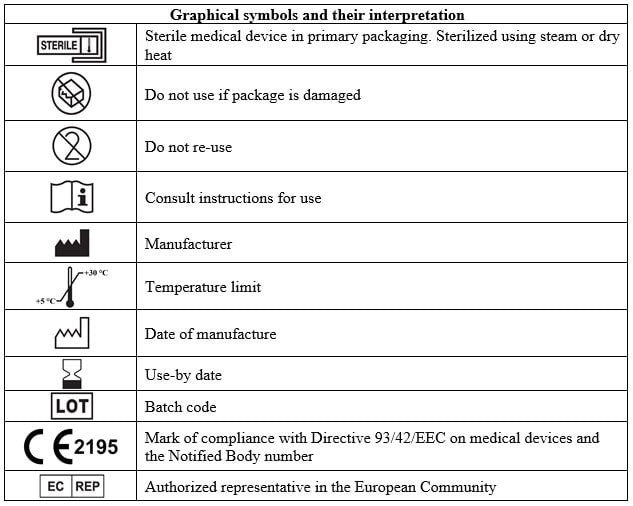
- Prior to application, verify the package integrity and check its shelf life. Do not use the device after the expiration date or if the package integrity is damaged.
- For single use only. Repeated use can lead to infection. Do not re-use.
- Dispose according to the requirements of local disposal regulations.
Storage conditions
Store in a place protected from sunlight at a temperature of +5 °С to +30 °С. Keep out of reach of children.
Shelf life
2 years. Shelf life is valid if the storage conditions are observed and the package is not damaged.
Authorized representative in the European Community
Diaco Biofarmaceutici S.R.L.
Via Flavia, 124, 34147, Trieste, Italy.
E-mail: info@diaco.it.
www.diaco.it
Name and address of the manufacturer
Yuria-pharm LLC, 10, M. Amosova Str., Kyiv, Ukraine, 03038.
Tel.: +38 (044) 275-92-42, +38 (044) 275-01-08.
E-mail: uf@uf.ua
www.uf.ua
Manufacturing site address: 108, Kobzarska Str., Cherkasy, Ukraine, 18030.
If you have any comments on the medical device or would like to give us feedback, please use the following options to contact us:
- email us at feedback@uf.ua;
- send a text message via Viber, Telegram or WhatsApp to the number: +38 (095) 275-33-01;
- call us at +38 (095) 275-33-01 or +38 (0800) 401-771 (charged in accordance with your operator’s tariff plan).
Date of last revision of instructions for use: 16.03.2023.
Version: 04.